 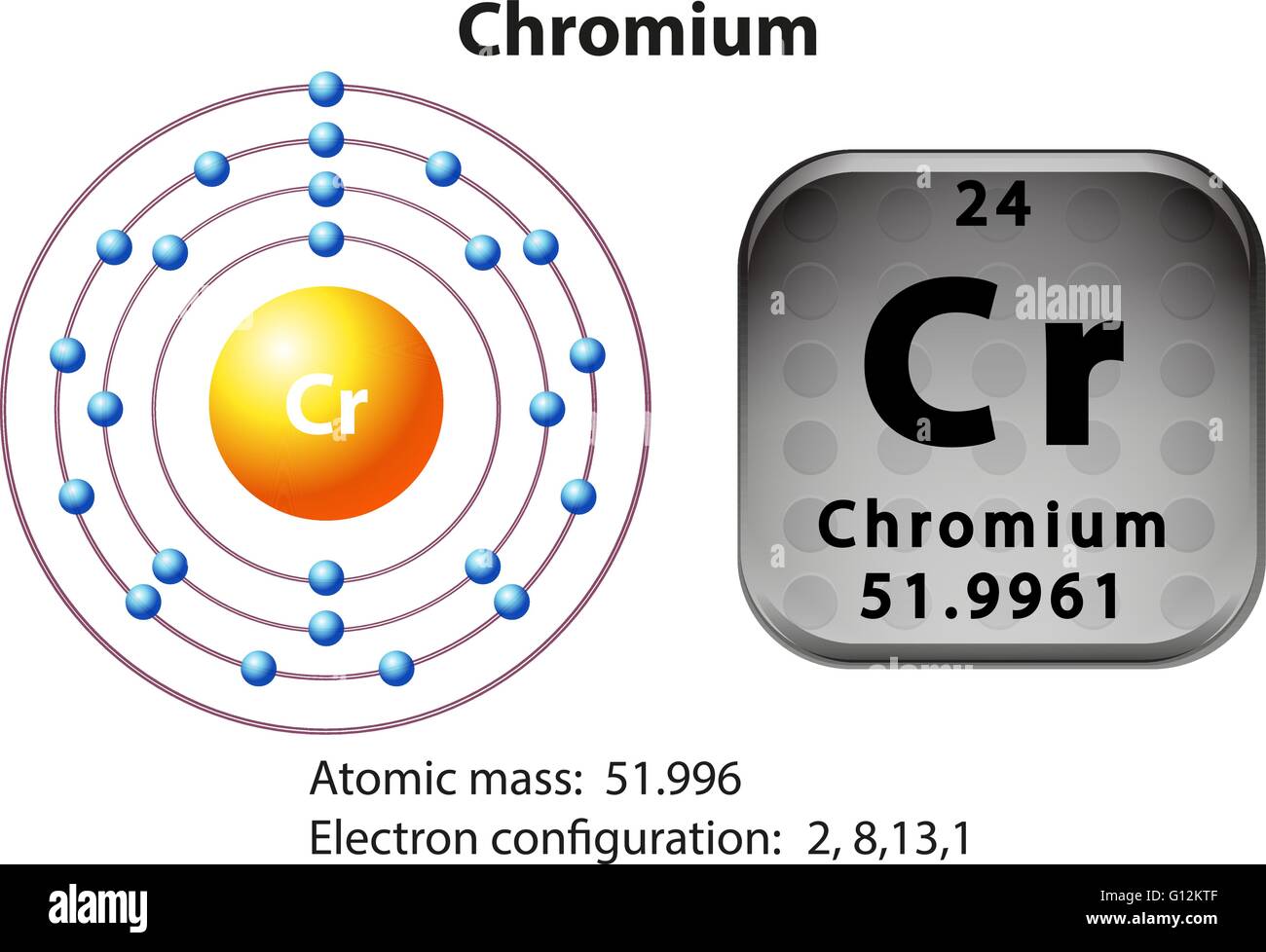
In all of these scenarios, the new redistribution of the electron is energetically favorable compared to the predicted configuration. You will see a similar situation where an #s# electron moves to a #d# sublevel with Molybdenum: So, in this case, an electron moves to #3d# and is unpaired, therefore maximizing the spin state. Since two opposite spins result in a total spin of #0#, maximizing this tends to require as many electrons in of same spin in different orbitals as possible. Hund's Rule: It is energetically favorable to maximize the spin state in a sublevel.The #3d# orbital is slightly lower in energy, and minimizing repulsions in the #4s# orbital by moving one of the #4s# electrons to a close-lying #3d# orbital minimizes the ground-state energy of chromium.This happens in Chromium, as one #4s# electron moves to the #3d# sublevel. This actually allows electrons to move between the sublevels if it is energetically favorable. Even though #4s# belongs to a higher main energy level than #3d#, the energy levels are similar. So why is this? It has to do with main the arrangement of sublevels in the electron cloud. However, the actual configuration of Chromium is: There is no hard and fast rule for this, but that is an explanation that correlates with experimental data.Looking at the periodic table, you would expect Chromium to have this configuration: Thus, electron pairing is favorable enough for Tungsten. The more the electron distribution is spread out, the less electron-pair repulsion there is, and thus the lower #Pi_c# is. However, Tungsten's #5d# and #6s# orbitals being larger than the #3d# and #4s# orbitals (respectively) spreads out the electron density enough that the pairing energy ( #Pi = Pi_c + Pi_e#) is small enough. The small-enough orbital size means that the electron density is not as spread out as it could be, which makes it favorable enough for a maximum total spin to give the most stable configuration.The minimization comes from having all unpaired electrons in the #3d# and #4s# ( #3d^5 4s^1#), rather than one electron pair in the #4s# ( #3d^4 4s^2#). The minimized coulombic repulsion energy #Pi_c# further stabilizes this configuration.The maximization comes from how there are #5# unpaired electrons, instead of just #4# ( #3d^4 4s^2#). The maximized exchange energy #Pi_e# stabilizes this configuration ( #3d^5 4s^1#).One explanation for Chromium, then, is that: The electron configuration of chromium, conventionally, would be displaystyle Ar4s23d4. The former is stabilizing and the latter is destabilizing, as shown below (suppose configuration 2 is at pairing energy #Pi = 0#): This is best exemplified by the transition metal chromium. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
